

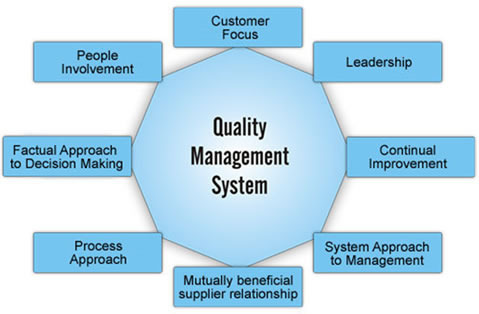
The pharmaceutical Quality Management System (QMS) holds significant importance as a crucial component, while encountering ongoing challenges during the audit process. From our perspective, the complexity lies in the application of the system, despite the presence of numerous knowledgeable individuals in the industry. Ensuring traceability and maintaining continuous monitoring are vital factors in achieving success. CPPL confidently claims that by obtaining the QMS Key and patiently waiting for six months, the result will be an infallible and strong system.
There are numerous approaches to GMP and pharmaceutical documentation.
1. Guideline Approach
2. Experience approach
3. Practical and productivity approach
These strategies exhibit variations among different manufacturers. Often, past audits play a significant role in the planning of future audits, as processes are tailored accordingly. Pharma QA, however, adopts a distinct methodology. We have observed that, apart from the auditors' perspective and interpretation of regulations, the guidelines provided by regulatory bodies are essentially identical. In order to adhere to regulatory guidelines worldwide, Pharma QA has devised a practical approach that enables the interpretation of rules. Each design created by Pharma QA ensures traceability and consistently yields commendable compliance results, as we firmly believe that justification for any noncompliance and traceability are of utmost importance.
Pharma QA offers quality integration services that enable pharmaceutical firms to fully deploy their systems and ensure compliance with global regulatory standards. The foundation of developing a Pharma QA system is a streamlined GMP Documentation.
1. A quality management system is designed by Pharma QA.
2. All systems and papers are provided by Pharma QA.
3. Pharma QA keeps an eye on every system.
4. Pharma QA Examines the systems.
5. Pharmaceutical Quality Assurance Establish a compliance level with requirements.